All published articles of this journal are available on ScienceDirect.
Predictive Modeling and Optimization of Compressive Strength for Cold In-Place Recycling Base Course Incorporating Ground Coal Bottom Ash and Calcium Chloride
Abstract
Introduction
Pavement rehabilitation faces increasing challenges from aging infrastructure, moisture damage, and traffic-induced fatigue. Cold In-Place Recycling (CIPR) provides a sustainable solution by reusing reclaimed asphalt pavement (RAP) and reducing reliance on virgin materials.
Methods
This study evaluated the compressive strength of CIPR base mixtures composed of crushed aggregate (CA) and RAP stabilized with Ordinary Portland Cement (OPC), ground coal bottom ash (GCBA), and calcium chloride (CaCl2). Three CA-RAP ratios (CA75RAP25, CA50RAP50, and CA25RAP75) were tested, with total stabilizer content fixed at 4% and CaCl2 varied from 0% to 3% of the total dry weight of the CA-RAP mixture. Unconfined Compressive Strength (UCS) tests were conducted at 1, 3, 7, 14, and 28 days to assess strength development. Response Surface Methodology (RSM) was employed to model and optimize the effects of CaCl2 and curing duration, producing statistically significant models (p < 0.05, R2 > 0.79).
Results
Results indicated that CaCl2 enhanced early-age strength, GCBA was most effective at moderate OPC levels, and UCS declined when CaCl2 exceeded 2%. The CA75RAP25 mix with 2% OPC and 2% GCBA achieved the highest UCS improvement.
Discussion
These findings highlight the interactive effects of stabilizer contents and curing duration, offering guidance for optimizing CIPR mixtures.
Conclusion
Incorporating GCBA and CaCl2 can enhance compressive strength, support sustainable reuse of industrial by-products, and improve the performance of pavement rehabilitation projects.
1. INTRODUCTION
Pavement deterioration caused by aging, moisture infiltration, and repeated traffic loading often leads to structural failures and a shortened service life, presenting substantial challenges for road maintenance [1, 2]. This often requires pavement rehabilitation, sometimes extending to base reconstruction to restore its load-bearing capacity and serviceability [3]. The most efficient approach to addressing these distresses is to undertake rehabilitation at an appropriate stage, thereby preventing further deterioration while optimizing resource utilization [4].
One of the available sustainable pavement rehabilitation methods is known as Cold In-Place Pavement Recycling (CIPR) [5-8]. This process involves scarifying the existing distressed pavement to a specific depth, typically between 50 mm and 150 mm, before mixing the reclaimed asphalt pavement (RAP) with stabilizers to form a new base course [9, 10]. Unlike conventional approaches requiring complete removal and replacement of pavement layers, CIPR reuses existing materials on-site, leading to reduced transportation costs, lower environmental impact, and energy savings [11, 12]. A life cycle assessment by Pakes et al. [13] demonstrated that CIPR reduces energy consumption and carbon dioxide emissions by approximately 23%, and water usage by 20%, when compared to traditional mill-and-overlay methods. Cost savings of up to 50% have also been reported, primarily due to decreased reliance on new aggregates and reduced haulage distances [14, 15].
The key constituents of CIPR mixtures are RAP and crushed aggregates (CA), which are recycled from aged pavements. RAP contributes to sustainability by reducing the need for virgin materials and lowering processing energy requirements [16]. However, RAP and CA alone often lack sufficient strength, requiring the addition of stabilizers to meet structural performance criteria [17].
In Malaysia, Ordinary Portland Cement (OPC) is commonly used as a stabilizer in CIPR applications [18]. However, its production contributes significantly to CO2 emissions, worsening global warming [19]. To promote sustainable construction, researchers have explored alternative cementitious materials that can partially or fully replace OPC while maintaining or improving mechanical properties.
One promising alternative is coal bottom ash (CBA), a by-product of coal combustion, which exhibits pozzolanic properties that improve the strength and durability of cement-treated materials [20, 21]. The effectiveness of CBA as a cement substitute depends on its fineness. Thus, it is finely ground to enhance its reactivity, forming ground coal bottom ash (GCBA). Additionally, calcium chloride (CaCl2) is often used as an accelerator to expedite hydration, leading to early strength gain in stabilized CIPR mixtures [22, 23].
Although the individual effects of GCBA and CaCl2 as stabilizers have been widely studied, limited research has investigated their combined influence in CIPR systems, especially under varying proportions of CA-RAP. This represents a critical gap, as the interaction between multiple stabilizers and recycled materials may produce synergistic or adverse effects that cannot be captured through isolated assessments.
Additionally, most previous studies rely on traditional trial-and-error approaches, which are both time-consuming and inefficient given the multi-variable nature of CIPR mix design. In contrast, Response Surface Methodology (RSM) has gained recognition as a useful statistical technique for modeling, analyzing, and optimizing engineering processes involving multiple interacting variables. RSM facilitates the development of predictive equations, identifies significant interaction effects, and enables the optimization of key performance parameters such as strength and durability. Recent studies in pavement engineering have successfully applied RSM to optimize mix compositions and predict mechanical properties in asphalt and recycled mixtures, highlighting its value in enhancing both material utilization and design efficiency [24-27]. Unlike conventional regression or single-variable statistical methods, RSM is specifically designed to evaluate interactions between variables and capture non-linear relationships with fewer experimental trials [28]. This makes it more efficient and accurate for multi-factor systems such as CIPR design mix, where stabilizer proportions and curing conditions interact in complex ways.
RSM has also been widely and successfully applied in other civil engineering applications. For example, it has been used to optimize operating parameters in wastewater treatment for COD/TOC removal in saline systems [29], to determine the optimum lime–fly ash dosage for improving CBR and strength in stabilized soils [30], and to enhance sustainable lateritic self-compacting concrete by incorporating waste laterite scraps as coarse aggregate [31].These diverse applications demonstrate the versatility and reliability of RSM in modeling complex, multi-variable systems, further supporting its use in CIPR design mix.
Therefore, this study applies RSM to investigate the combined effects of OPC, GCBA, and CaCl2 on the compressive strength of CIPR mixtures. Laboratory experiments were conducted to generate data for model development, with the aim of establishing a statistically significant model that captures the interaction effects between stabilizer contents and curing conditions, enabling accurate predictions and efficient mix optimization. By integrating GCBA and CaCl2 into cement-stabilized CIPR mixtures and modeling their combined effects using RSM, this study moves beyond previous research focused on other stabilizers or trial-and-error mix designs. The approach provides insights for optimizing sustainable CIPR bases through material innovation and advanced statistical modeling, supporting sustainable pavement rehabilitation by incorporating industrial waste and applying data-driven methods to enhance performance while reducing environmental impact.
2. MATERIALS
The pavement base course design mix incorporated two primary materials: CA and RAP, both of which play a crucial role in ensuring the durability and load-bearing capacity of the structure. In enhancing the mechanical performance and long-term stability of the base course, stabilizers were introduced. These included GCBA, OPC, and CaCl2, each contributing unique properties that could improve strength, cohesion, and resistance to environmental factors. The combination of these materials aimed to optimize the structural integrity and sustainability of the pavement system.
2.1. Crushed Aggregate (CA)
The CA was sourced from Kajang Rock Quarry, Selangor, Malaysia. Gradation was controlled to comply with the Public Works Department (PWD) Malaysia specifications for pavement applications. The material was sieved to meet the required size distribution, as detailed in Table 1. The corresponding particle size distribution curve is shown in Fig. (1).
2.2. Recycle Asphalt Pavement (RAP)
The RAP was obtained from a milling operation at a road construction site in Selangor. It was selected to promote material recycling and reduce dependence on virgin aggregates. The RAP was thoroughly blended with CA to form the structural base layer of the CIPR mixtures. This combination aimed to improve the mechanical performance, durability, and cost-effectiveness of the base course while promoting the reuse of waste materials in road construction.
2.3. Design Mix Composition and Properties of CA-RAP
The combinations of CA and RAP were varied in 25% increments, and each mix was evaluated for aggregate quality through tests including shape, abrasion, and impact resistance. The mixed compositions and test results are summarized in Tables 2 and 3, respectively.
| BS Sieve Size (mm) | PWD Requirement Cumulative % Passing | Cumulative % Passing |
|---|---|---|
| 50.0 | 100 | 100 |
| 37.5 | 85-100 | 100 |
| 28.0 | 70-100 | 96.9 |
| 20.0 | 60-90 | 78.8 |
| 10.0 | 40-65 | 52.5 |
| 5.0 | 30-55 | 44.4 |
| 2.0 | 20-40 | 23.8 |
| 0.425 | 10-25 | 15.8 |
| 0.075 | 2-10 | 2.7 |
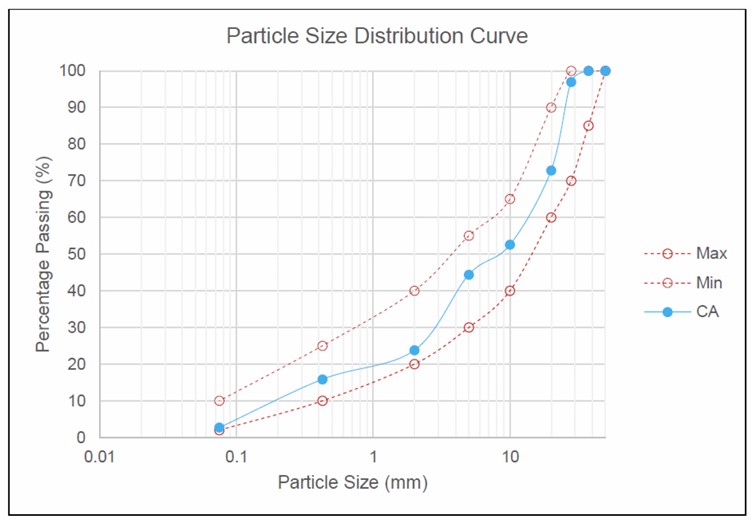
Particle size distribution curve of crushed aggregate.
| Mix Designation | Percentage of CA (%) | Percentage of RAP (%) |
|---|---|---|
| CA75RAP25 | 75 | 25 |
| CA50RAP50 | 50 | 50 |
| CA25RAP75 | 25 | 75 |
| Aggregate Test | Result (%) | PWD Requirement (%) | Reference |
|---|---|---|---|
| Particle Shape Analysis (Combine value of Elongation and Flakiness Index) (%) | CA75RAP25: 17.38 |
<25 | BS 812-105.2: 1990 and MS30 |
| CA50RAP50: 20.04 |
|||
| CA25RAP75: 22.5 |
|||
| Los Angeles (LA) abrasion value (%) | CA75RAP25: 28.68 |
<45 | ASTM C 131-96 |
| CA50RAP50: 35.21 |
|||
| CA25RAP75: 38.43 |
|||
| Aggregate Impact Value, AIV (%) | CA75RAP25: 15.99 |
<25 | BS 812-112: 1990 |
| CA50RAP50: 17.34 |
|||
| CA25RAP75: 19.55 |
2.4. Stabilizer
This study utilized OPC of CEM Type I, alongside raw CBA obtained from the Jana Manjung Power Plant in Perak. Due to its high silica content, CBA is believed to have great potential as a pozzolanic material. However, its natural form is not suitable for direct application in cementitious mix because of its high porosity and relatively large particle size. In improving its reactivity, CBA must be finely ground so that the material retained on a No. 325 sieve (44 μm) does not exceed 5%, as highlighted by Jaturapitakkul & Cheerarot [32]. In this study, CBA was ground to achieve a finer particle size, ensuring it passed through a 45 μm sieve, thereby producing GCBA with enhanced pozzolanic properties.
Furthermore, CaCl2 was also introduced as an accelerator. The CaCl2 used was a flake industrial-grade material with a minimum purity of 74%, known for its ability to expedite cement hydration and significantly improve the early strength development of cementitious mix.
All percentages of the stabilizers were based on the total dry weight of the CA-RAP sample mixture. The total stabilizer content, comprising OPC and GCBA, was fixed at 4%, with CaCl2 ranging from 0% to 3%. The control specimen was stabilized solely with OPC at 4%. This 4% content was identified as the optimal dosage for effective stabilization during construction, as it provided sufficient early strength development and was based on site data. The combinations of 2% OPC and 2% GCBA, 3% OPC and 1% GCBA, and 4% OPC with 0% GCBA were evaluated in this study. The combination of 1% OPC and 3% GCBA was excluded due to its poor performance in the earlier strength test. Tables 4 and 5 show the percentage of stabilizer used in this study and their properties, respectively.
| Percentage of Stabilizer | OPC (%) | GCBA (%) | CaCl2 (%) |
|---|---|---|---|
| 4% | 2 | 2 | 0,1,2,3 |
| 3 | 1 | 0,1,2,3 | |
| 4 | 0 | 1,2,3 | |
| Control Specimen | 4 | 0 | 0 |
| Stabilizer | Specific Gravity, Gs | pH Value | Chemical Composition |
|---|---|---|---|
| OPC | 2.93 | 9.85 | C, O, Na, Mg, Al, Si, P. Ca, Fe, S, K |
| GCBA | 2.56 | 14.31 | C, O, Mg, Al, Si. Ca, S, K |
| CaCl2 | - | 6.35 | Ca, Cl |
3. METHODS
This section outlines the experimental testing and statistical modeling procedures used to evaluate the compressive strength performance of the CIPR mixtures. The workflow includes laboratory UCS testing followed by Response Surface Methodology (RSM) modeling and statistical analysis.
3.1. Research Framework
The study framework consists of three main phases: unconfined compressive strength (UCS) testing, data modeling using RSM, and statistical validation. The relationship between the independent variables (OPC, GCBA, CaCl2 content, and curing time) and the response variable (UCS) was analyzed to develop predictive models. The overall experimental process, including design, testing, and modeling, is summarized in Fig. (2).
3.2. Unconfined Compressive Strength Test
The UCS test is a key indicator of the mechanical integrity of stabilized CIPR mixtures and was conducted in accordance with ASTM D1633. Cylindrical specimens (100 mm diameter) were prepared from the CA-RAP mixtures and compacted to the target density using a static compactor. Three specimens from each design mix to allow averaging of the results were prepared and cured for 1, 3, 7, 14, and 28 days at room temperature (±25 °C).
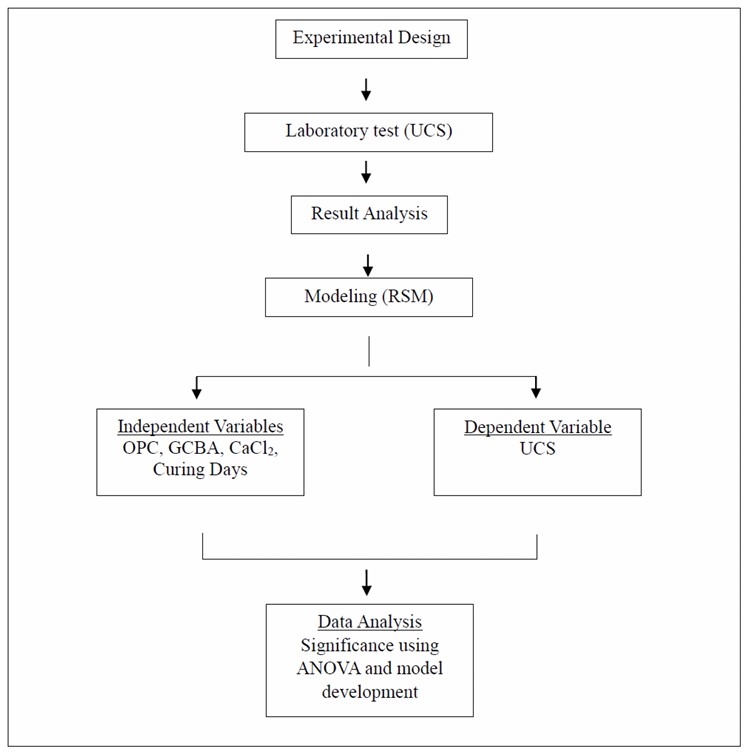
Three-phase framework.
After curing, specimens were subjected to axial compressive loading at a constant rate of 153 kN/min until failure. The peak load was recorded and used to compute the UCS value (in MPa).
UCS results for all three CA-RAP compositions under different stabilizer combinations are presented in Tables 6-8.
| OPC:GCBA:CaCl2 ratio (%) | UCS Value (MPa) | ||||
|---|---|---|---|---|---|
| Day 1 | Day 3 | Day 7 | Day 14 | Day 28 | |
| 2:2:0 | 2.24 | 2.51 | 2.54 | 2.81 | 3.93 |
| 3:1:0 | 3.40 | 3.43 | 3.55 | 3.95 | 4.75 |
| 4:0:0 | 3.46 | 3.58 | 3.76 | 4.04 | 4.78 |
| 2:2:1 | 2.45 | 2.62 | 4.13 | 4.19 | 4.37 |
| 3:1:1 | 3.31 | 3.71 | 4.15 | 4.73 | 5.18 |
| 4:0:1 | 3.25 | 3.75 | 4.17 | 4.60 | 4.99 |
| 2:2:2 | 2.49 | 2.95 | 3.07 | 3.14 | 3.99 |
| 3:1:2 | 3.04 | 3.97 | 4.04 | 4.08 | 5.01 |
| 4:0:2 | 3.09 | 4.08 | 4.38 | 4.51 | 4.83 |
| 2:2:3 | 1.98 | 2.15 | 2.39 | 2.54 | 2.77 |
| 3:1:3 | 2.58 | 2.81 | 3.06 | 3.10 | 3.37 |
| 4:0:3 | 2.45 | 2.68 | 3.40 | 3.52 | 3.69 |
| OPC:GCBA:CaCl2 Ratio (%) | UCS Value (MPa) | ||||
|---|---|---|---|---|---|
| Day 1 | Day 3 | Day 7 | Day 14 | Day 28 | |
| 2:2:0 | 2.57 | 3.16 | 3.45 | 3.67 | 4.53 |
| 3:1:0 | 2.97 | 3.95 | 4.04 | 4.75 | 4.80 |
| 4:0:0 | 3.56 | 3.97 | 4.29 | 4.79 | 5.10 |
| 2:2:1 | 3.40 | 3.52 | 3.64 | 4.00 | 4.51 |
| 3:1:1 | 3.51 | 3.83 | 4.59 | 4.74 | 5.83 |
| 4:0:1 | 3.86 | 3.91 | 4.40 | 4.86 | 5.93 |
| 2:2:2 | 3.16 | 3.40 | 3.60 | 3.62 | 4.76 |
| 3:1:2 | 3.44 | 3.83 | 4.39 | 4.73 | 5.13 |
| 4:0:2 | 3.84 | 4.19 | 4.25 | 4.81 | 5.52 |
| 2:2:3 | 2.20 | 2.38 | 2.73 | 2.88 | 4.14 |
| 3:1:3 | 3.03 | 3.49 | 3.70 | 3.96 | 4.89 |
| 4:0:3 | 3.26 | 3.31 | 3.40 | 4.39 | 4.50 |
| OPC:GCBA:CaCl2 ratio (%) | UCS Value (MPa) | ||||
|---|---|---|---|---|---|
| Day 1 | Day 3 | Day 7 | Day 14 | Day 28 | |
| 2:2:0 | 2.94 | 3.00 | 3.37 | 4.00 | 4.74 |
| 3:1:0 | 3.49 | 3.96 | 4.27 | 4.64 | 4.72 |
| 4:0:0 | 3.25 | 3.82 | 4.21 | 4.61 | 4.76 |
| 2:2:1 | 3.05 | 3.20 | 3.62 | 4.48 | 4.54 |
| 3:1:1 | 3.20 | 3.53 | 4.41 | 4.70 | 4.90 |
| 4:0:1 | 3.51 | 3.52 | 4.46 | 4.69 | 5.04 |
| 2:2:2 | 2.67 | 2.71 | 3.02 | 3.41 | 3.79 |
| 3:1:2 | 3.27 | 3.57 | 3.88 | 4.25 | 4.30 |
| 4:0:2 | 3.35 | 3.55 | 3.96 | 4.23 | 4.98 |
| 2:2:3 | 2.37 | 2.43 | 2.78 | 2.87 | 3.30 |
| 3:1:3 | 2.83 | 3.28 | 3.68 | 3.92 | 4.16 |
| 4:0:3 | 3.09 | 3.27 | 3.43 | 3.88 | 4.48 |
| CA-RAP Mix Ratio | Independent Variables | Dependent Variable | |||
|---|---|---|---|---|---|
| Fixed Factor | Factor A | Factor B | |||
| OPC (%) | GCBA (%) | CaCl2 (%) | Curing Days | Response | |
| CA75RAP25 | 2,3,4 | 0,1,2 | 0,1,2 | 1,3,7,14,28 | UCS |
| CA50RAP50 | 2,3,4 | 0,1,2 | 0,1,2 | 1,3,7,14,28 | UCS |
| CA25RAP75 | 2,3,4 | 0,1,2 | 0,1,2 | 1,3,7,14,28 | UCS |
| Stabilizer | F-value | p-value | R2 | Adjusted R2 | Predicted R2 | Model Type | ||
|---|---|---|---|---|---|---|---|---|
| OPC (%) | GCBA (%) | CaCl2 (%) | ||||||
| 2 | 2 | 0,1,2 | 8.16 | 0.0037 | 0.8193 | 0.7188 | 0.5839 | Quadratic |
| 3 | 1 | 0,1,2 | 30.63 | <0.0001 | 0.8362 | 0.8089 | 0.7568 | Linear |
| 4 | 0 | 0,1,2 | 45.48 | <0.0001 | 0.7979 | 0.7642 | 0.6809 | Linear |
3.3. Model Design and Variable Selection
RSM was employed to model the influence of key factors on UCS performance, as it provides a systematic approach for analyzing variable interactions and non-linear effects while minimizing the number of experimental trials required. Among the four potential input variables - OPC, GCBA, CaCl2, and curing duration - only two (CaCl2 content and curing duration) were selected as independent variables in the RSM models to simplify analysis and avoid model overfitting. The OPC and GCBA ratios were held constant for each design set.
Three stabilizer combinations were tested:
• 2% OPC + 2% GCBA
• 3% OPC + 1% GCBA
• 4% OPC + 0% GCBA
These combinations were applied across all CA-RAP ratios with the presence of CaCl2. The response variable was UCS. Table 9 summarizes the experimental variables.
3.4. Regression Modeling and ANOVA
The effects of CaCl2 content (A) and curing duration (B) on the UCS of CIPR mixtures were modeled using RSM. Three polynomial models of increasing complexity were developed: a linear model (Eq. 1), a two-factor interaction model (Eq. 2), and a quadratic model (Eq. 3):
Y=β0 + β1A + β2B (1)
Y=β0 + β1 A + β2 B + β12 A B (2)
Y=β0 +β1 A +β2 B +β12 AB +β11 A2 +β22 B2 (3)
Where;
Y = response (UCS, MPa),
A , B = independent variables (CaCl2 and curing duration)
β = model coefficient
The significance of each model was evaluated using analysis of variance (ANOVA). A model was considered statistically significant if the p-value was < 0.05. Model performance was validated using R2, adjusted R2, and predicted R2 values.
4. RESULTS ANALYSIS
This section presents the modeling results for the UCS of CIPR mixtures incorporating varying combinations of OPC, GCBA, and CaCl2 across three CA-RAP mix ratios: CA75RAP25, CA50RAP50, and CA25RAP75. For each mix type, regression models were developed and validated using ANOVA. The model type (linear or quadratic) was selected based on p-value, F-value, and overall fit statistics, including R2, adjusted R2, and predicted R2.
4.1. CA75RAP25
In the CA75RAP25 mix, as presented in Table 10, the quadratic model for the composition containing 2% OPC and 2% GCBA exhibited more complex interactions and nonlinear effects. This nonlinearity suggests that the combined influence of the stabilizers and curing time had a synergistic impact on UCS, which could not be accurately represented by a simple linear model. In contrast, the other mixtures demonstrated simpler relationships between input variables and UCS, where changes in UCS followed a more predictable linear pattern.
The UCS prediction models for this mix are presented in Eqs. (4-6).
• Eq. (4): 2% OPC + 2% GCBA
UCS = 1.9735 + 1.3880A + 0.1086B - 0.0057AB - 0.5833A - 0.0015B2
• Eq. (5): 3% OPC + 1% GCBA
UCS = 3.2985 + 0.1061A + 0.0579B
• Eq. (6): 4% OPC + 0% GCBA
UCS = 3.4139 + 0.1257A + 0.0514B
Where:
A: CaCl2 content (%)
B: Curing duration (days)
Figure 3 illustrates the 3-D response surface models for the CA75RAP25 mix, depicting UCS variations with respect to CaCl2 content and curing duration at fixed OPC: GCBA ratios.
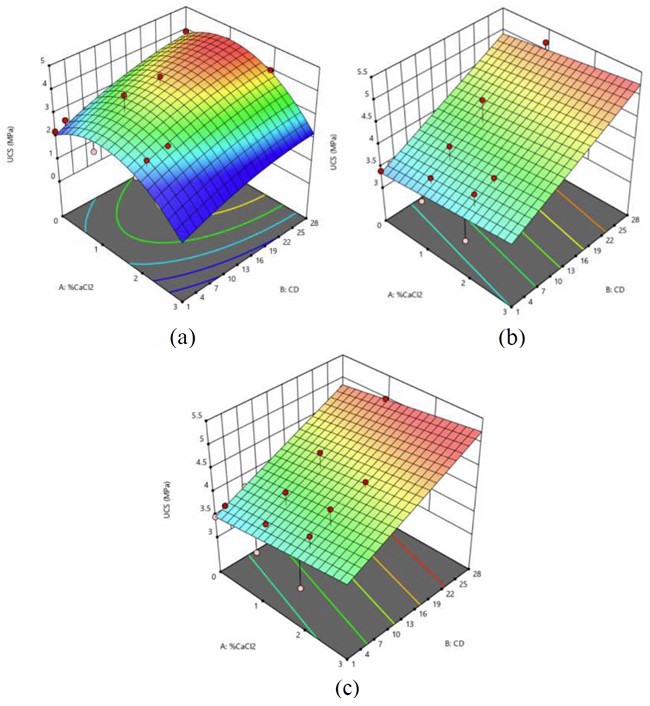
3-D Model Analysis for CA75RAP25 Design Mix Consists of a) 2% OPC + 2% GCBA, b) 3% OPC + 1% GCBA, and c) 4% OPC with various percentages of CaCl2.
The UCS showed an initial improvement with increasing CaCl2 content in the 2% OPC and 2% GCBA mix. This trend is likely due to the accelerating effect of CaCl2, which promotes early hydration and strength gain by facilitating the rapid formation of calcium silicate hydrate (C-S-H). However, at higher concentrations, CaCl2 may lead to excessive heat of hydration and formation of microcracks, which can negatively affect UCS.
| Stabilizer | F-value | p-value | R2 | Adjusted R2 | Predicted R2 | Model Type | ||
|---|---|---|---|---|---|---|---|---|
| OPC (%) | GCBA (%) | CaCl2 (%) | ||||||
| 2 | 2 | 0,1,2 | 45.13 | <0.0001 | 0.8827 | 0.8361 | 0.8226 | Linear |
| 3 | 1 | 0,1,2 | 18.13 | 0.0002 | 0.9097 | 0.8595 | 0.6157 | Quadratic |
| 4 | 0 | 0,1,2 | 71.50 | <0.0001 | 0.9226 | 0.9097 | 0.8457 | Linear |
For the 3% OPC and 1% GCBA model, UCS showed a steady increase with rising CaCl2 content. The higher OPC content likely provided a greater amount of reactive calcium silicates, enhancing the formation of C-S-H when activated by CaCl2. The relatively lower GCBA content may have reduced pozzolanic dilution, allowing OPC to dominate the strength development process.
In the 4% OPC mix (with 0% GCBA), a significant rise in UCS was also observed with increasing CaCl2 content. This further supports the role of CaCl2 in accelerating cement hydration, particularly when sufficient OPC is available. The absence of GCBA in this mix may have resulted in fewer competing pozzolanic reactions, allowing for a more direct OPC-CaCl2 interaction that favors rapid strength gain.
These findings suggest that CaCl2 enhances UCS primarily through hydration acceleration, and that its effectiveness depends on the proportion of OPC present. While GCBA contributes long-term pozzolanic reactivity, its presence in higher amounts may reduce early strength unless optimally balanced with OPC and activators.
4.2. CA50RAP50
Table 11 summarizes the CA50RAP50 mix, where both linear and quadratic models revealed varying degrees of interaction between the proportions of OPC and GCBA. Notably, the mix containing 3% OPC and 1% GCBA exhibited more complex, nonlinear behavior compared to other combinations. These results indicate that the balance between OPC and GCBA plays a critical role in governing the strength development process.
The UCS prediction models for this mix are presented in Eqs. (7-9).
• Eq. (7): 2% OPC + 2% GCBA
UCS = 2.9894 + 0.1167A + 0.0529B
• Eq. (8): 3% OPC + 1% GCBA
UCS = 3.1155 + 0.6879A + 0.1514B + 0.0008AB - 0.2983A2 - 0.0029B2
• Eq. (9): 4% OPC + 0% GCBA
UCS = 3.7231 + 0.0906A + 0.0633B
Where:
A: CaCl2 content (%)
B: Curing duration (days)
The 3-D response surface models for the CA50RAP50 mix are shown in Figure 4, demonstrating UCS variations with respect to CaCl2 content and curing duration at fixed OPC: GCBA ratios.
In the model with 2% OPC and 2% GCBA, UCS increased with higher CaCl2 content, reflecting the positive role of CaCl2 in accelerating hydration and promoting early formation of strength-contributing compounds such as C-S-H. This effect is particularly beneficial in mixtures where GCBA's pozzolanic reaction is slower and benefits from early calcium availability.
In the model with 3% OPC and 1% GCBA, UCS initially increased with CaCl2 content but declined at higher concentrations. This trend suggests that while moderate CaCl2 addition enhances hydration and strength gain, excessive levels may cause rapid heat evolution and microcracking, offsetting the early strength gains. Furthermore, the relatively low GCBA content may limit secondary pozzolanic reactions that could otherwise help buffer this rapid setting.
The model with 4% OPC (and 0% GCBA) exhibited a continuous increase in UCS as CaCl2 dosage rose, mirroring the trend observed in the CA75RAP25 mix. In this case, the high cement content provided a sufficient supply of reactive phases, allowing CaCl2 to effectively accelerate hydration without adverse effects. The absence of GCBA might have simplified the reaction dynamics, reduced variability, and contributed to more predictable strength improvement.
Overall, these findings reinforce the role of CaCl2 as a hydration accelerator and highlight the importance of optimizing its dosage based on the mix composition.
4.3. CA25RAP75
In the CA25RAP75 mix, as illustrated in Table 12, the analysis revealed that quadratic models for all mixtures exhibited significant interactions and non-linear effects. These effects are likely attributed to the increased variability introduced by the high RAP content, which tends to create more heterogeneous mixtures. The presence of a larger proportion of aged binders and aggregates in RAP may have contributed to unpredictable behavior, especially when interacting with varying levels of stabilizers. Consequently, the mix's response to changes in OPC, GCBA, and CaCl2 became more sensitive and nonlinear.
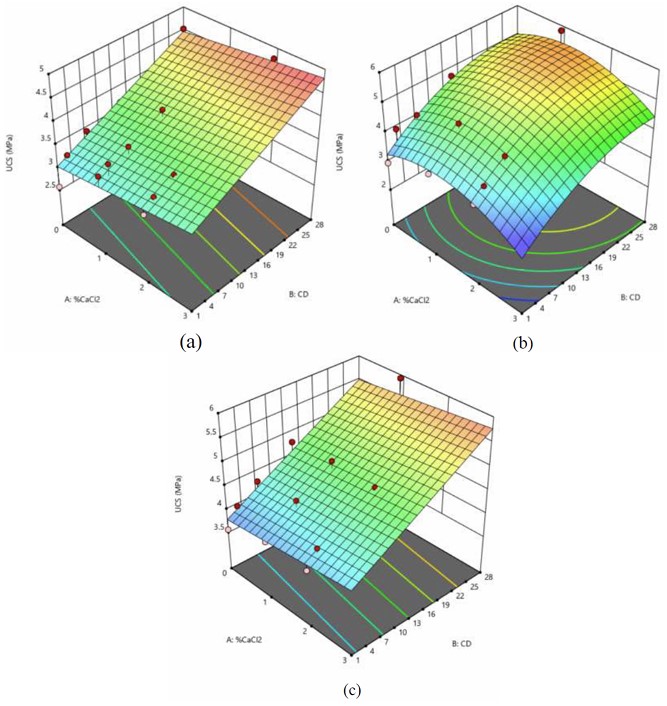
3-D model analysis for CA50RAP50 design mix consists of a) 2% OPC + 2% GCBA, b) 3% OPC + 1% GCBA, and c) 4% OPC with Various Percentages of CaCl2.
| Stabilizing Agent | F-value | p-value | R2 | Adjusted R2 | Predicted R2 | Model Type | ||
|---|---|---|---|---|---|---|---|---|
| OPC (%) | GCBA (%) | CaCl2 (%) | ||||||
| 2 | 2 | 0,1,2 | 65.58 | <0.0001 | 0.9733 | 0.9584 | 0.9231 | Quadratic |
| 3 | 1 | 0,1,2 | 23.57 | <0.0001 | 0.9291 | 0.8896 | 0.7836 | Quadratic |
| 4 | 0 | 0,1,2 | 27.63 | <0.0001 | 0.9388 | 0.9049 | 0.8083 | Quadratic |
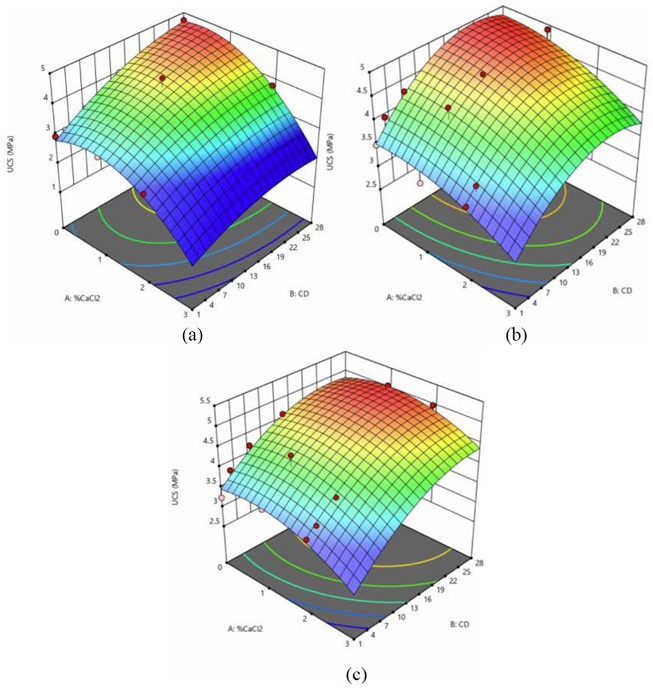
Model analysis for CA25RAP75 design mix consists of a) 2% OPC + 2% GCBA, b) 3% OPC + 1% GCBA, and c) 4% OPC with various percentages of CaCl2.
The UCS prediction models for this mix are presented in Eqs. (10-12).
• Eq. (10): 2% OPC + 2% GCBA
UCS = 2.6770 + 0.7188A +0.1249B - 0.0129AB - 0.4130A2 - 0.0018B2
• Eq. (11): 3% OPC + 1% GCBA
UCS = 3.3674 + 0.0678A + 0.1467B - 0.0022AB - 0.1122A2 - 0.0034B2
• Eq. (12): 4% OPC + 0% GCBA
UCS = 3.3358 + 0.2354A + 0.1249B + 0.0046AB - 0.1714A2 - 0.0025B2
Where:
A: CaCl2 content (%)
B: Curing duration (days)
Figure 5 shows the 3-D response surface models for the CA25RAP75 mix, which demonstrate a consistent trend across different mix compositions.
In the mix containing 2% OPC and 2% GCBA, UCS increased with rising CaCl2 content up to 2%, after which it declined. A similar trend was observed in the 3% OPC + 1% GCBA and 4% OPC mixtures, where UCS peaked at a CaCl2 content of approximately 1–2% before decreasing.
This behavior suggests that while CaCl2 effectively accelerates hydration and enhances early strength development, excessive dosages may trigger adverse effects such as rapid setting, internal stress buildup, and microcracking, particularly in mixtures with high RAP content. The aged binder in RAP may hinder effective bonding between newly formed hydration products and aggregate surfaces, making the matrix more susceptible to premature stiffening or shrinkage caused by excess CaCl2. In addition, the less compatible combination of OPC and GCBA in high-RAP mixtures may have limited cohesion and compaction quality, amplifying the sensitivity to CaCl2 dosage when the aged binder further reduces bonding efficiency and disrupts stabilizer interaction.
These findings highlight the importance of optimizing the balance between OPC, GCBA, and CaCl2 in high-RAP mixtures. Excessive accelerator content in such mixtures may compromise long-term performance despite initial gains, highlighting the need for careful dosage control when designing CIPR mixtures with high RAP proportions.
Overall, the developed RSM models demonstrated strong statistical reliability, with R2 values consistently exceeding 0.79 across all CA-RAP ratios and stabilizer combinations. This confirms that the models effectively captured the influence of CaCl2 content and curing duration on UCS performance, providing sufficient accuracy to support practical mix design optimization.
5. DISCUSSION
The results demonstrated that both CaCl2 content and curing duration significantly influenced the UCS of CIPR mixtures across all CA-RAP ratios. Linear models showed that incremental increases in CaCl2 generally enhanced UCS, particularly at early curing stages. Similar findings were reported by Shon et al. [22], who observed that CaCl2 accelerated cement hydration and improved early strength in stabilized bases. However, quadratic trends revealed that strength gains plateaued or declined beyond a threshold of approximately 2%, consistent with Salain [23], who reported that excessive CaCl2 led to premature setting and microcracking in cementitious systems. Collectively, these results confirm that while CaCl2 is effective for accelerating hydration and early strength development, overdosing may compromise long-term performance. This trend was most evident in high-RAP mixtures (CA25RAP75), where the presence of aged binder and a reduced cementitious matrix increased sensitivity to rapid hydration, hindering the formation of a well-integrated microstructure and making the mix more vulnerable to the adverse effects of higher CaCl2 dosages.
The 3D response surface models further illustrated these patterns, showing steady UCS increases with CaCl2 addition, particularly in 4% OPC mixtures. This suggests that higher OPC content enhances hydration kinetics and supports the formation of dense calcium silicate hydrate (C-S-H) networks. However, when CaCl2 dosage exceeded 2%, UCS declined across all mix types, highlighting the need to balance early strength acceleration with long-term microstructural stability.
The contribution of GCBA, by contrast, diminished in mixtures with high OPC and CaCl2 contents. Mixtures with little or no GCBA often achieved higher UCS, suggesting that the slower pozzolanic reaction of GCBA was overshadowed by the faster hydration of OPC under accelerated conditions. This outcome aligns with Argiz et al. [21, 17], who noted that bottom ash requires extended curing to manifest its pozzolanic activity, and Alosta et al. [20], who emphasized that proper treatment and curing are essential to enhance its reactivity in blended systems.
From a practical perspective, the developed RSM models provide a valuable tool for field engineers to predict strength development under different curing periods and stabilizer dosages. They also highlight the risks of overdosing CaCl2, which may deliver short-term strength benefits but could compromise long-term durability in CIPR applications.
6. LIMITATIONS AND FUTURE WORKS
This study was limited to UCS evaluations under controlled laboratory conditions. To establish field applicability, future research should examine long-term durability, moisture susceptibility, and environmental performance of optimized CIPR formulations. Full-scale field trials are also recommended to validate the predictive accuracy of the developed RSM models under actual construction and service conditions. Furthermore, future work should assess the economic feasibility of the proposed CIPR mixtures, considering the costs of incorporating GCBA and CaCl2 and potential savings from using recycled materials. Such analysis would provide practical guidance for real-world implementation and support informed decision-making in sustainable pavement rehabilitation projects.
CONCLUSION
This study investigated the compressive strength performance of CIPR mixtures stabilized with OPC, GCBA, and CaCl2 across three CA-RAP ratios. A combination of laboratory UCS testing and RSM modeling enabled the evaluation and prediction of strength development under varying stabilizer dosages and curing durations.
Key findings are as follows:
1. GCBA contributed most effectively to UCS when paired with moderate OPC content (2–3%). Its influence diminished in mixtures with higher OPC or CaCl2 contents, where rapid cement hydration dominated strength development.
2. CaCl2 enhanced early-age UCS, especially within the first 7 curing days. However, dosages above 2% reduced strength, likely due to rapid setting, internal stress buildup, and microcracking.
3. The CA75RAP25 mixture with 2% OPC and 2% GCBA achieved the highest UCS improvement (up to ~20% higher than the control), demonstrating the synergistic balance between cement hydration and pozzolanic reactivity.
4. The RSM models developed in this study provide a practical framework for optimizing CIPR mix designs, supporting both performance improvement and sustainability in pavement rehabilitation.
Overall, the findings demonstrate the potential of combining OPC, GCBA, and CaCl2 to enhance the mechanical performance of CIPR base mixtures.
AUTHORS' CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: E.S.: Conceptualization; A.I.: Investigation; M.I.J., R.P.J.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| CA | = Crushed Aggregate |
| RAP | = Reclaimed Asphalt Pavement |
| CIPR | = Cold In-Place Recycling |
| OPC | = Ordinary Portland Cement |
| CBA | = Coal Bottom Ash |
| GCBA | = Ground Coal Bottom Ash |
| CaCl2 | = Calcium Chloride |
| UCS | = Unconfined Compressive Strength |
| RSM | = Response Surface Methodology |
AVAILABILITY OF DATA AND MATERIALS
All data, experimental results, and materials used in this study were generated by the authors through laboratory work conducted at Universiti Teknologi MARA (UiTM). The data supporting the findings of the article will be available from the corresponding author [E.S] upon reasonable request.
ACKNOWLEDGEMENTS
The authors would like to acknowledge the support and facilities provided by Universiti Teknologi MARA (UiTM) Shah Alam and UiTM Cawangan Pulau Pinang, which were instrumental in conducting this study.


